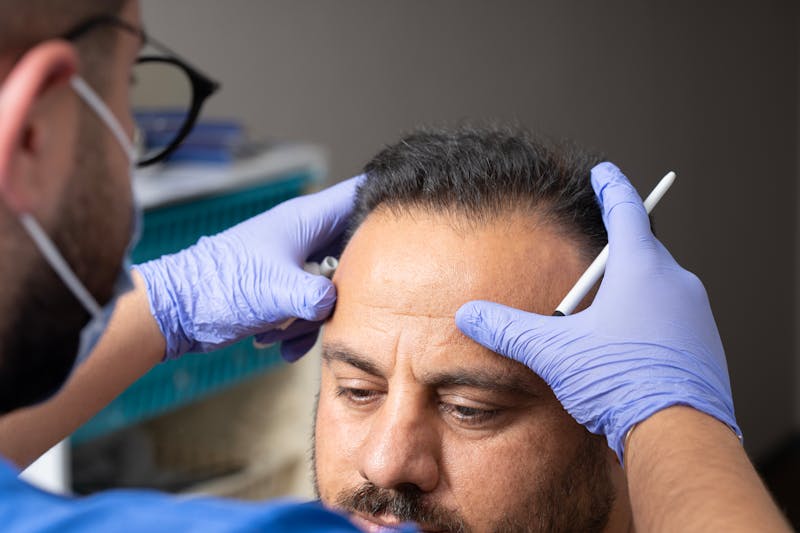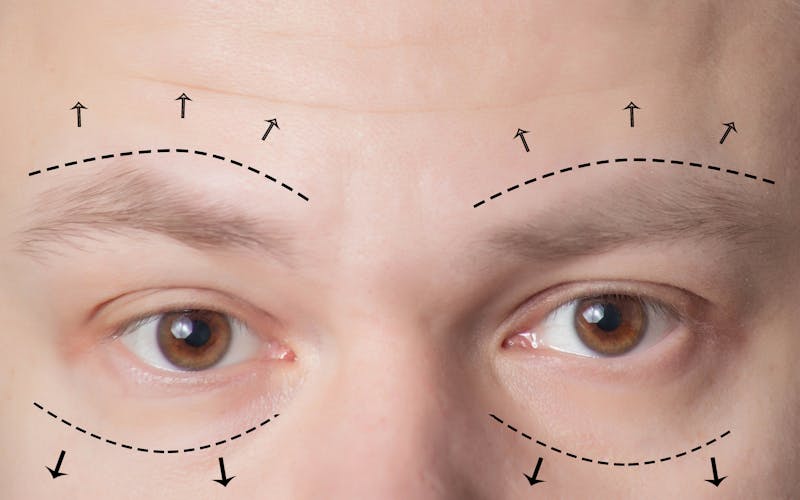
How Do I Get My Upper Blepharoplasty To Last?
I see many patients who come into my office who have gone to amazing surgeons for their upper blepharoplasties, but they're missing one key component to ensuring longevity from their procedure: the brow. When you treat the upper eyelid, you have to evaluate and often treat ...
View More